- Blog
- Service def system dodge
- Companionlink software reviews
- How to see hidden files with flashfxp
- Solidworks toolbox read only
- Nord vpn download
- Serious sam 1 binky
- Skyrim dragonborn dlc pc
- 108 posturas de yoga pdf
- Mister retro set five
- Latest driverpack solution offline
- Tutorial how to instal element 3d v2-2
- Ivms 4200 client not opening
- Microsoft visual studio 2017 free download
- Nexus plugin playing same sound
- Full pokemon game for android
- Adobe photoshop 5-0 update
- Inputmapper ds4 windows 10
- Allwinner a33 specs
- Korg pa1x pro software download
- Inductive automation ignition costs
- Pbp3 ecoli uniprot
- Rangbaaz web series complete all episode 480p
- When did batman arkham knight come out
- Colony survival wiki
- Titan quest atlantis builds
- Dance music 320kbps

isolates, molecular typing and phylogenetic analysis, overview Clonal relationships among Citrobacter spp. AmpC molecular characterization shows high diversity into blaCMY and blaACT genes from Citrobacter and Enterobacter species, respectively, with low clonality among them. A high percentage of resistance to ampicillin is detected (57 %), observing the AmpC phenotype in 22 isolates (31 %) and the extended spectrum beta-lactamase (ESBL) phenotype in 3 isolates.
50 human faecal samples are obtained, and 70 Enterobacteriaceae bacteria are isolated: 44 Escherichia coli, 4 Citrobacter braakii, 9 Citrobacter freundii, 8 Enterobacter cloacae, 1 Proteus mirabilis, 1 Proteus vulgaris, 1 Klebsiella oxytoca, 1 Serratia sp. Beta-lactamases and other resistance mechanisms are characterized in Enterobacteriaceae isolates recovered from healthy human faecal samples, focusing on the ampC beta-lactamase genes. Phenotype and genotype of the AmpC-positive strains. Docking studies reveal that the azolylthioacetamides, which have the broadest inhibitory activity, coordinate to the ZnII ion(s) preferentially via the triazole moiety, while other moieties interact mostly with the conserved active site residues No inhibition by N-(1,3-benzothiazol-2-yl)-2-sulfanyl]acetamide, N-(1,3-benzothiazol-2-yl)-2-sulfanyl]acetamide, N-phenyl-2-sulfanyl]acetamide, and N-phenyl-2-sulfanyl]acetamide. Our results provide evidence that structure-activity relationship studies for the purposes of drug discovery must consider both β-lactamases and penicillin-binding proteins as targets.Development of diaryl-substituted azolylthioacetamides as metallo-beta-lactamase-1 enzyme inhibitors, overview no inhibition by N-(1,3-benzothiazol-2-yl)-2-sulfanyl]acetamide, N-phenyl-2-sulfanyl]acetamide, N-(1,3-benzothiazol-2-yl)-2-sulfanyl]acetamide, N-(1,3-benzothiazol-2-yl)-2-sulfanyl]acetamide, N-phenyl-2-sulfanyl]acetamide, and N-phenyl-2-sulfanyl]acetamide, poor inhibition by N-(1,3-benzothiazol-2-yl)-2-sulfanyl]acetamide, 2-sulfanyl]-N-phenylacetamide, N-(1,3-benzothiazol-2-yl)-2-sulfanyl]acetamide, N-phenyl-2-sulfanyl]acetamide, N-(1,3-benzothiazol-2-yl)-2-sulfanyl]acetamide, and N-phenyl-2-sulfanyl]acetamide development of eighteen diaryl-substituted azolylthioacetamides as metallo-beta-lactamase enzyme inhibitors, overview. We then show that avibactam derivatives retain β-lactamase inhibitory properties but also exhibit considerable antimicrobial activity against clinically relevant bacteria via targeting penicillin-binding proteins. A kinetic analysis of key active-site mutants for class A β-lactamase CTX-M-15 allows us to propose a validated mechanism for avibactam-mediated β-lactamase inhibition including a unique role for S130, which acts as a general base. To understand the molecular basis and spectrum of inhibition by avibactam, we provide structural and mechanistic analysis of the compound in complex with important class A and D serine β-lactamases. The novel diazabicyclooctane (DBO) avibactam inhibits a wider range of serine β-lactamases than has been previously observed with clinical β-lactamase inhibitors. An analysis of the NDM-1 active site in these structures reveals key features important for the informed design of novel inhibitors of NDM-1 and other metallo-β-lactamases.

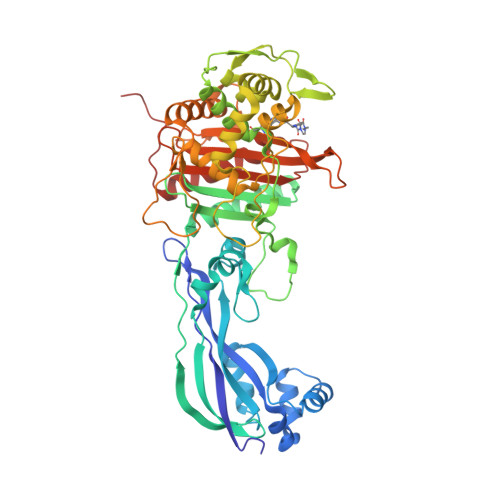
The structural details underpinning the broad-spectrum resistance of NDM-1 was further investigated by analysis of the protein in complex with hydrolyzed β-lactams as well as bound to the inhibitor L-captopril. Furthermore, NDM-1 localizes to the bacterial outer-membrane by sucrose density gradient centrifugation. pneumoniae holo-NDM-1 revealed an expansive active site, which we propose leads to a broader β-lactam substrate specificity. The 2.1Å resolution crystal structure of K. Recently, a novel resistance factor known as the New Delhi Metallo-β-Lactamase-1 has been found to confer enteric pathogens such as Escherichia coli and Klebsiella pneumoniae with nearly complete resistance to all β-lactams. This thesis aims to better understand the molecular details governing extended-spectrum β-lactamase mediated β-lactam resistance, and to gain insights into inhibition of these emerging resistance factors. To protect the clinical utility of the β-lactams it is essential to understand the structural basis for this resistance.
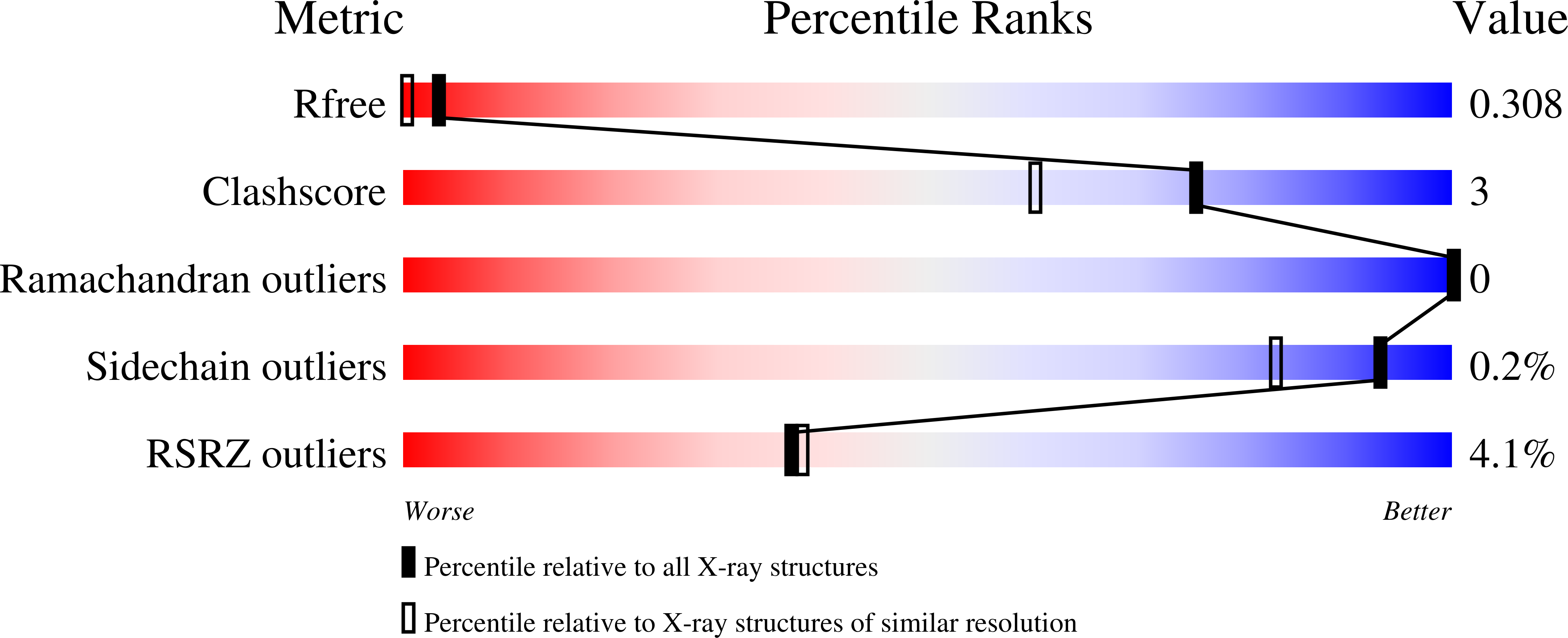
However, bacteria have gained resistance to all major classes of β-lactams. The most widespread class of human antibacterials is the β-lactams that target the transpeptidase activity of penicillin-binding proteins, which are responsible for cross-linking the peptidoglycan cell-wall. Bacterial diseases have an enormous impact on human health.
- Blog
- Service def system dodge
- Companionlink software reviews
- How to see hidden files with flashfxp
- Solidworks toolbox read only
- Nord vpn download
- Serious sam 1 binky
- Skyrim dragonborn dlc pc
- 108 posturas de yoga pdf
- Mister retro set five
- Latest driverpack solution offline
- Tutorial how to instal element 3d v2-2
- Ivms 4200 client not opening
- Microsoft visual studio 2017 free download
- Nexus plugin playing same sound
- Full pokemon game for android
- Adobe photoshop 5-0 update
- Inputmapper ds4 windows 10
- Allwinner a33 specs
- Korg pa1x pro software download
- Inductive automation ignition costs
- Pbp3 ecoli uniprot
- Rangbaaz web series complete all episode 480p
- When did batman arkham knight come out
- Colony survival wiki
- Titan quest atlantis builds
- Dance music 320kbps
